Execute Phase III Trials at Global Scale, Unprecedented Speed
Unify thousands of patients and hundreds of sites on a single, AI-powered infrastructure. Accelerate time to database lock and regulatory submission without compromising data integrity or compliance.
Carelane is a unified, AI-powered infrastructure for global Phase III trials. Carelane is a unified, AI-powered infrastructure for global Phase III trials. By integrating EDC, ePRO, eCOA and eConsent into a single source of truth, Carelane eliminates silos and slashes onboarding timelines. AI-driven query resolution cleans datasets 90% faster, accelerating database lock and regulatory submission. With 50% faster protocol amendments and 80% less site burden, Carelane delivers the scale and precision needed for market exclusivity.
Why Global Phase III Trials Face Exponential Friction
Pivotal trials require massive coordination across countries, sites, and systems. Traditional infrastructure buckles under this complexity, resulting in crippling data silos, delayed queries, and painful site fatigue.
Your pivotal trial infrastructure should observational tools, not interventional overhead.
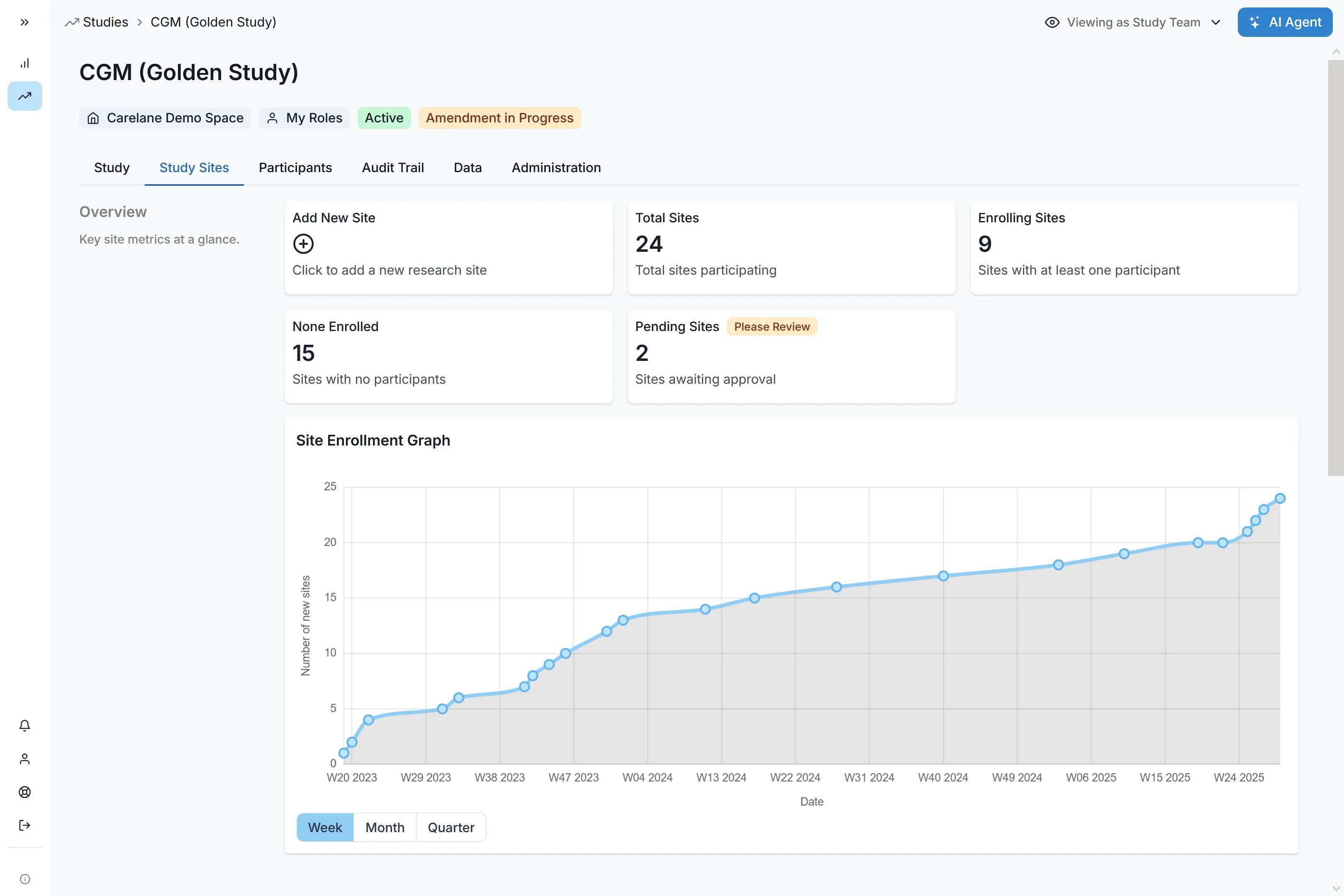
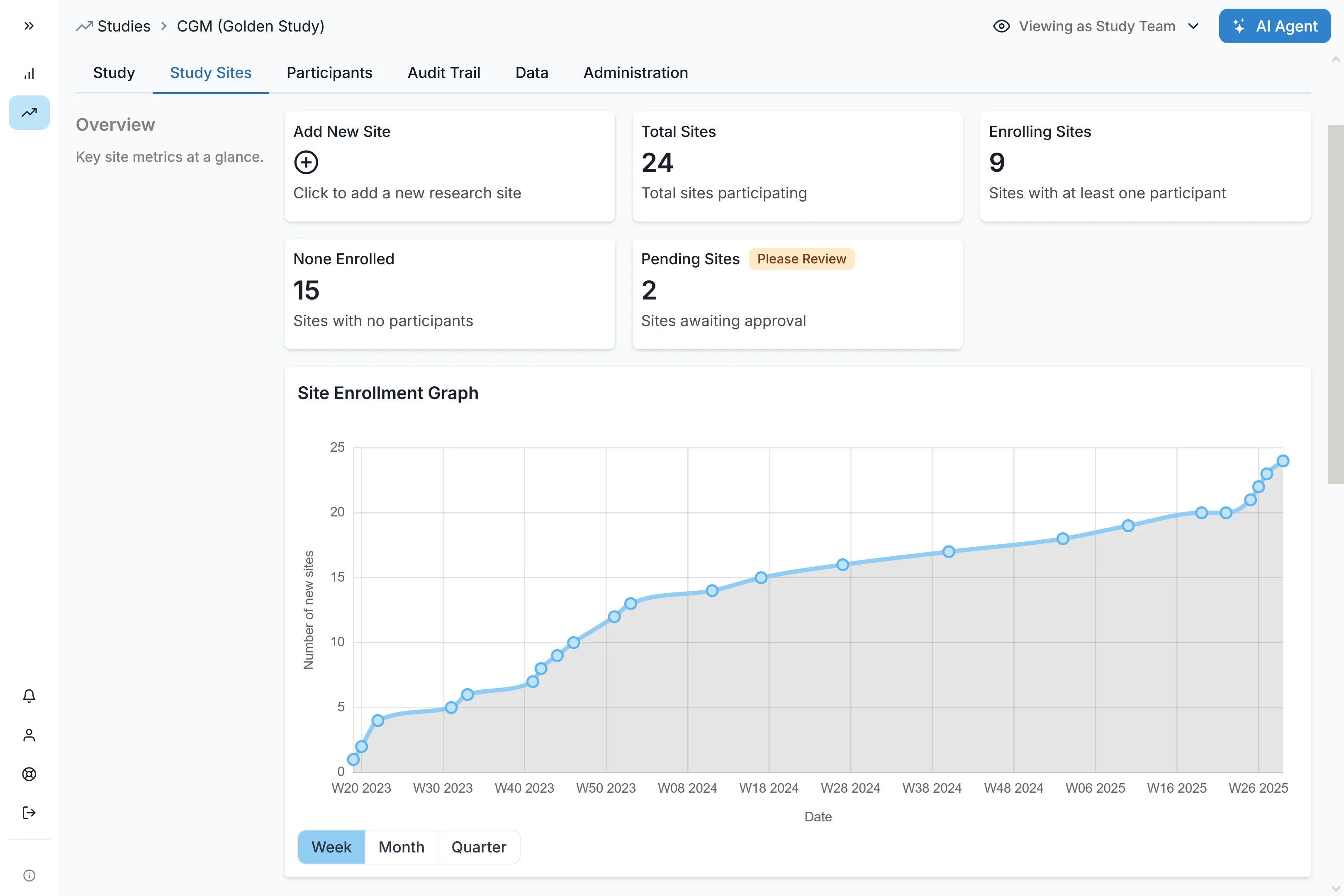
Unified Infrastructure Built for Global Scale
Carelane operates as your single source of truth. By unifying your trial ecosystem and injecting human-in-the-loop AI, we eliminate manual friction and slash Phase III timelines.
Lightning-Fast Global Site Activation
Deploy to hundreds of sites across multiple continents in a fraction of the time. Automated self-registration workflows and integrated feasibility assessments reduce site selection from 10 weeks to 1 week, accelerating your global footprint.
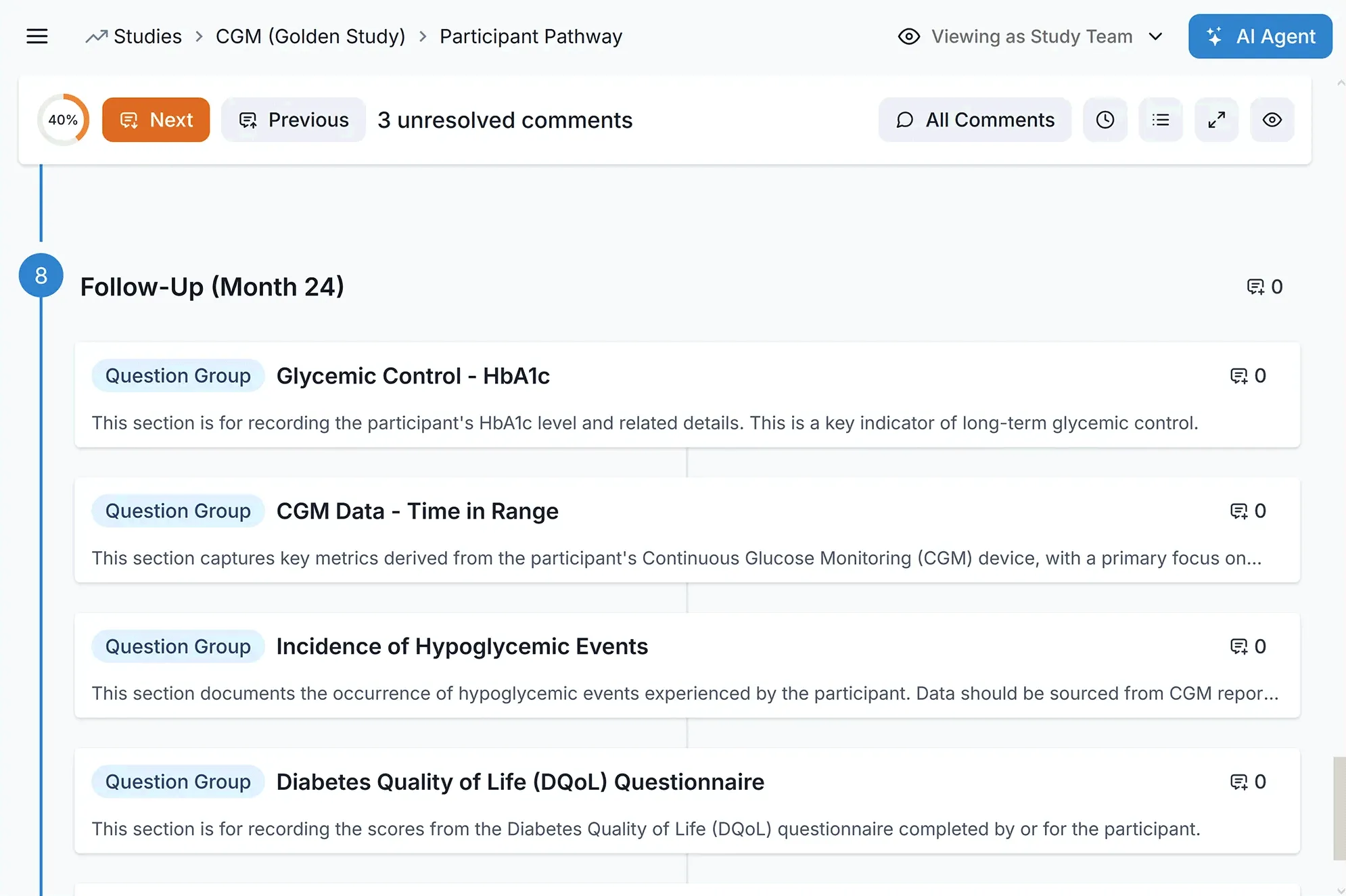
AI-Powered Query Management at Scale
Keep massive datasets pristine in real-time. Carelane’s intelligent edit checks prevent bad data at the source, while AI-assisted resolution slashes query response times from weeks to days, keeping you on a hyper-fast track to database lock.
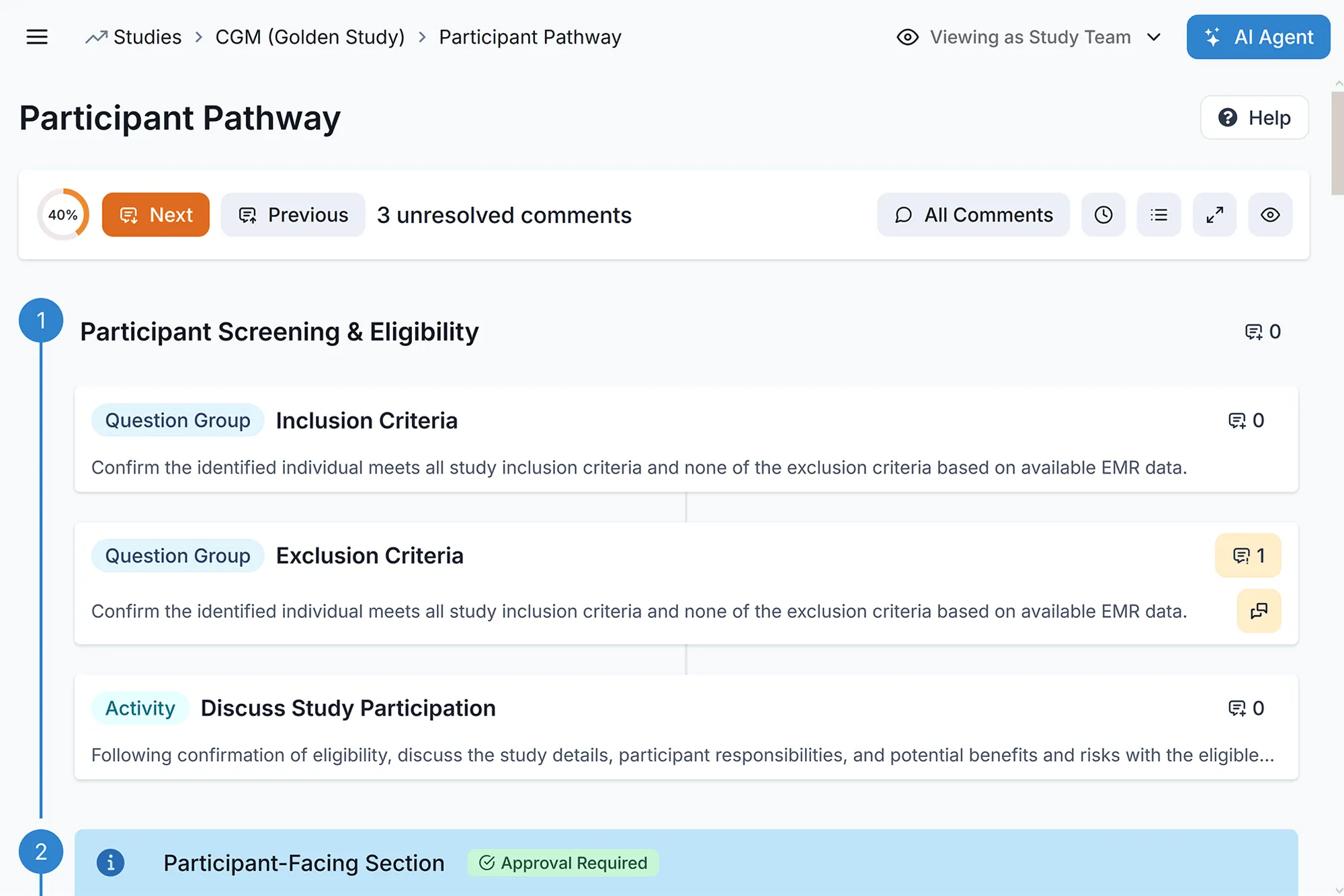
Unified Patient Engagement (ePRO & eConsent)
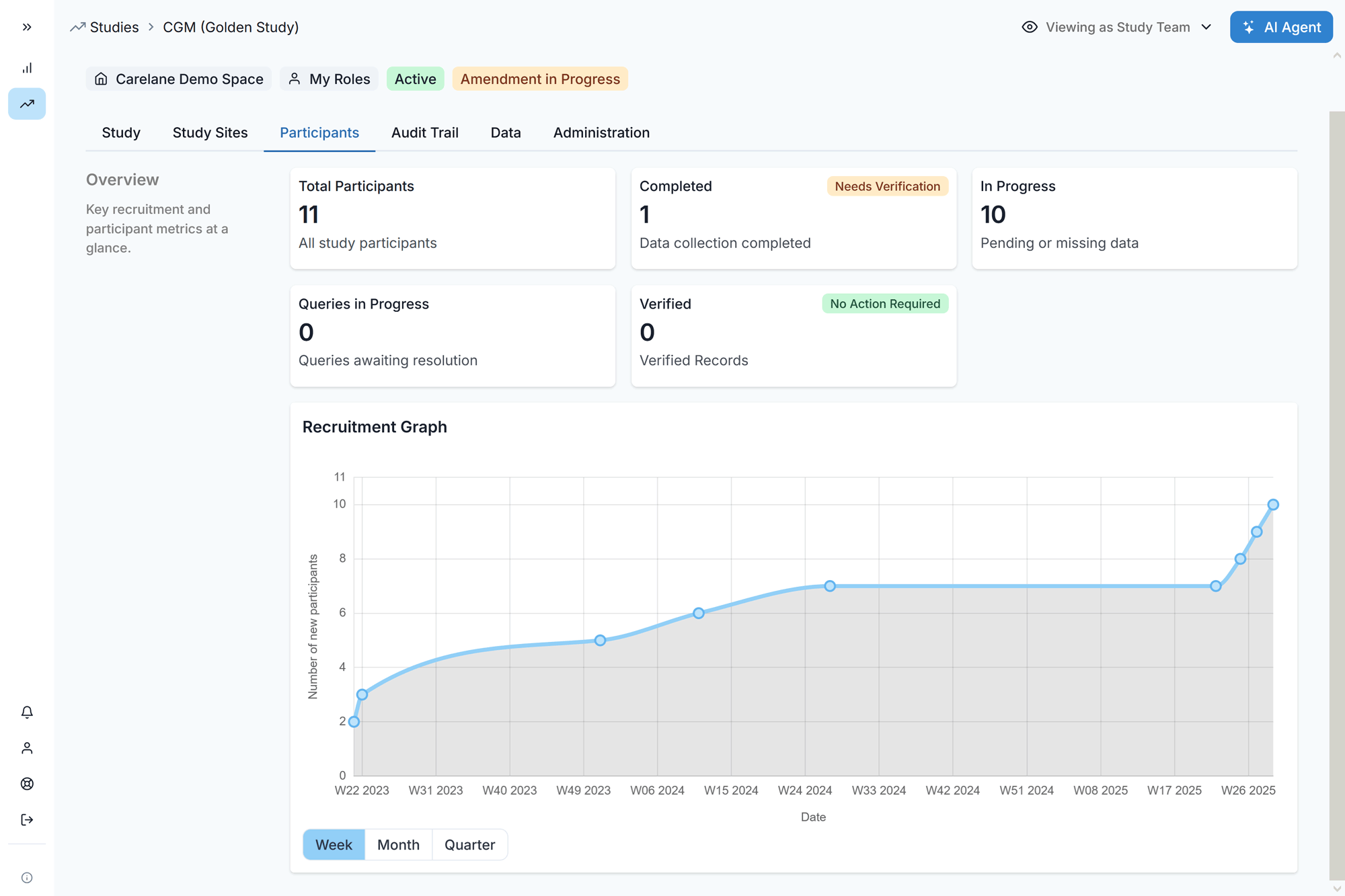
Drive long-term retention with a seamless patient experience. Streamlined consents and intuitive, mobile-first ePRO workflows integrate directly into your EDC. This unified approach reduces practitioner burden and enables you to double patient enrollment and retention.
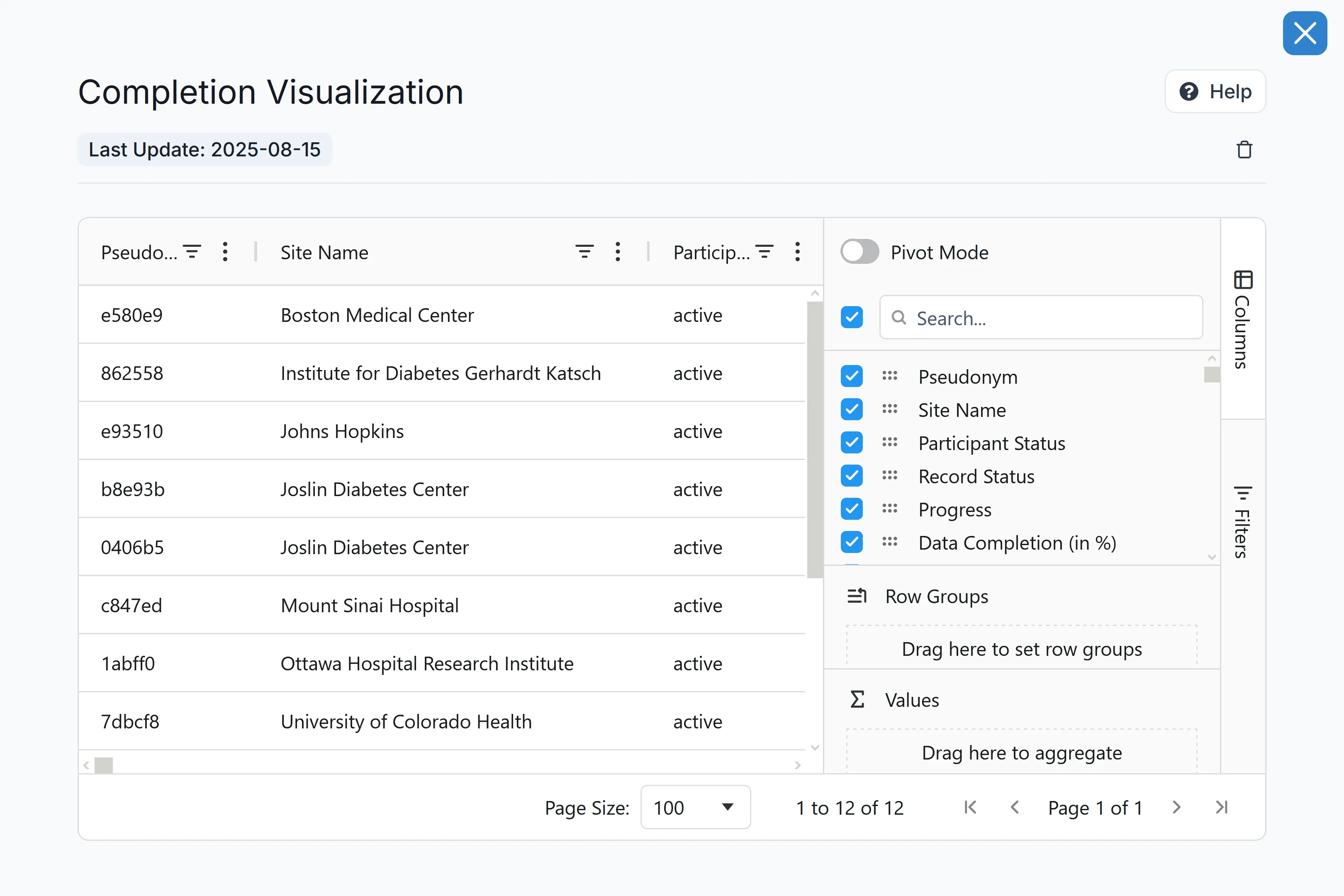
Automated Site Support & Global Oversight
Managing hundreds of global sites shouldn't require an army of support staff. Carelane's centralised, real-time data visibility and integrated communication tools drastically reduce site support efforts from 20 person-days to just 1 person-day. You get complete global oversight, and your sites get the instant support they need to keep enrolling and treating patients without delay.
Proven at Scale Across the Globe
Global sponsors and CROs trust Carelane’s AI-driven platform to manage their most critical, high-volume Phase III trials with flawless speed and precision.
- 70-90% reduction in query management time across 100+ sites
- Complete elimination of ePRO-to-EDC reconciliation delays
- Database lock achieved significantly ahead of the traditional industry baseline
The Strategic Value of Speed to Submission
In Phase III, every day delayed is a day of lost market exclusivity and delayed patient access. Modern pivotal trials require infrastructure that accelerates the finish line.
The Race to Regulatory Submission
Speed is the ultimate competitive advantage. A unified platform that auto-reconciles data and resolves queries in real-time ensures your clinical study report and regulatory filings happen months earlier.
Eradicating
Vendor Silos
Regulatory bodies expect pristine, easily auditable data trails. Operating on a single platform with zero-knowledge encryption and RBAC eliminates the compliance risks associated with transferring data between 5 different legacy vendors.
Human-in-the-loop AI Adoption
Manual data management cannot scale to meet modern Phase III demands. AI is no longer optional; it is the new standard for sponsors who want to remain competitive, drastically reducing CRF design times and query backlogs.
Ready to try Carelane?
The new standard for Sponsors, CROs and Institutions to run faster trials. Instant adoption. Human-in-the-loop AI. Zero compliance risk.