Accelerate Phase II Trials with Unrivalled Speed and Signal Clarity
Move from proof-of-concept to pivotal trials faster. Unify EDC, ePRO, eCOA, and site workflows on an AI-driven platform designed to slash study timelines, eliminate data silos, and deliver immediate efficacy insights.
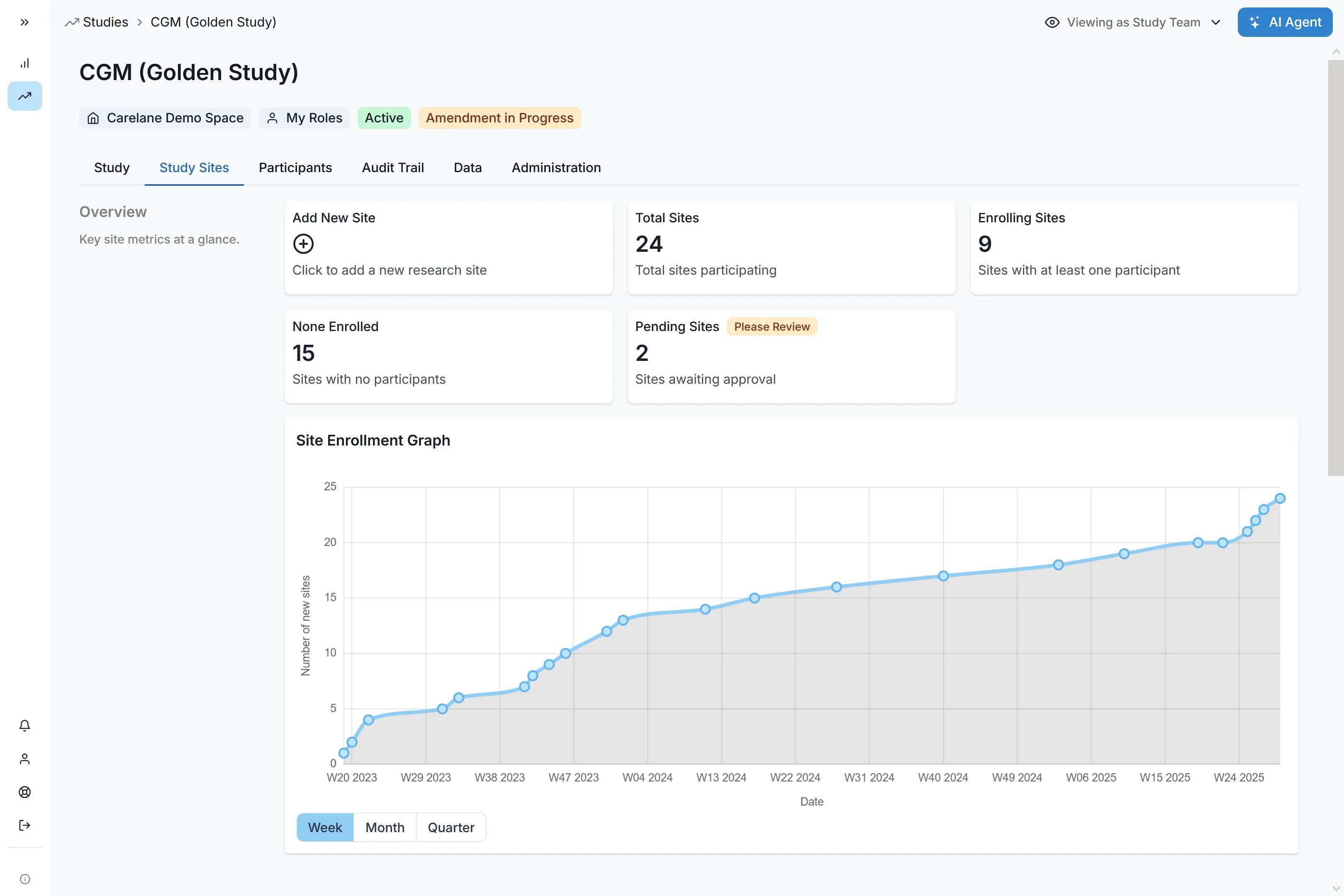
Carelane is an AI-driven platform built to accelerate Phase II proof-of-concept trials by unifying EDC, ePRO, eCOA and site workflows. Carelane eliminates mid-stage bottlenecks via automated site self-registration, reducing onboarding from 10 weeks to 1 week, and AI-assisted query resolution that cuts data cleaning time by 90%. By providing a single source of truth for real-time efficacy signals and 50% faster protocol amendments, Carelane ensures Go/No-Go decisions and Phase III readiness are achieved with unmatched speed and data clarity.
Why Phase II Trials Get Bogged Down
Phase II is the critical bridge to commercial viability. Yet, fragmented legacy systems, sluggish site activation, and manual data cleaning often obscure early efficacy signals and drain capital.
Your clinical infrastructure should illuminate efficacy, not hide it behind administrative friction.
Speed by Default. Quality by Design.
Carelane replaces disjointed legacy tech with a unified, high-speed ecosystem, empowering you to execute Phase II trials with unprecedented efficiency and real-time clarity.

Automated Site Self-Registration
Scale your sites in record time. Automated self-registration workflows and integrated feasibility assessments reduce the site selection and onboarding process from 10 weeks to just 1 week, letting you start recruiting immediately.
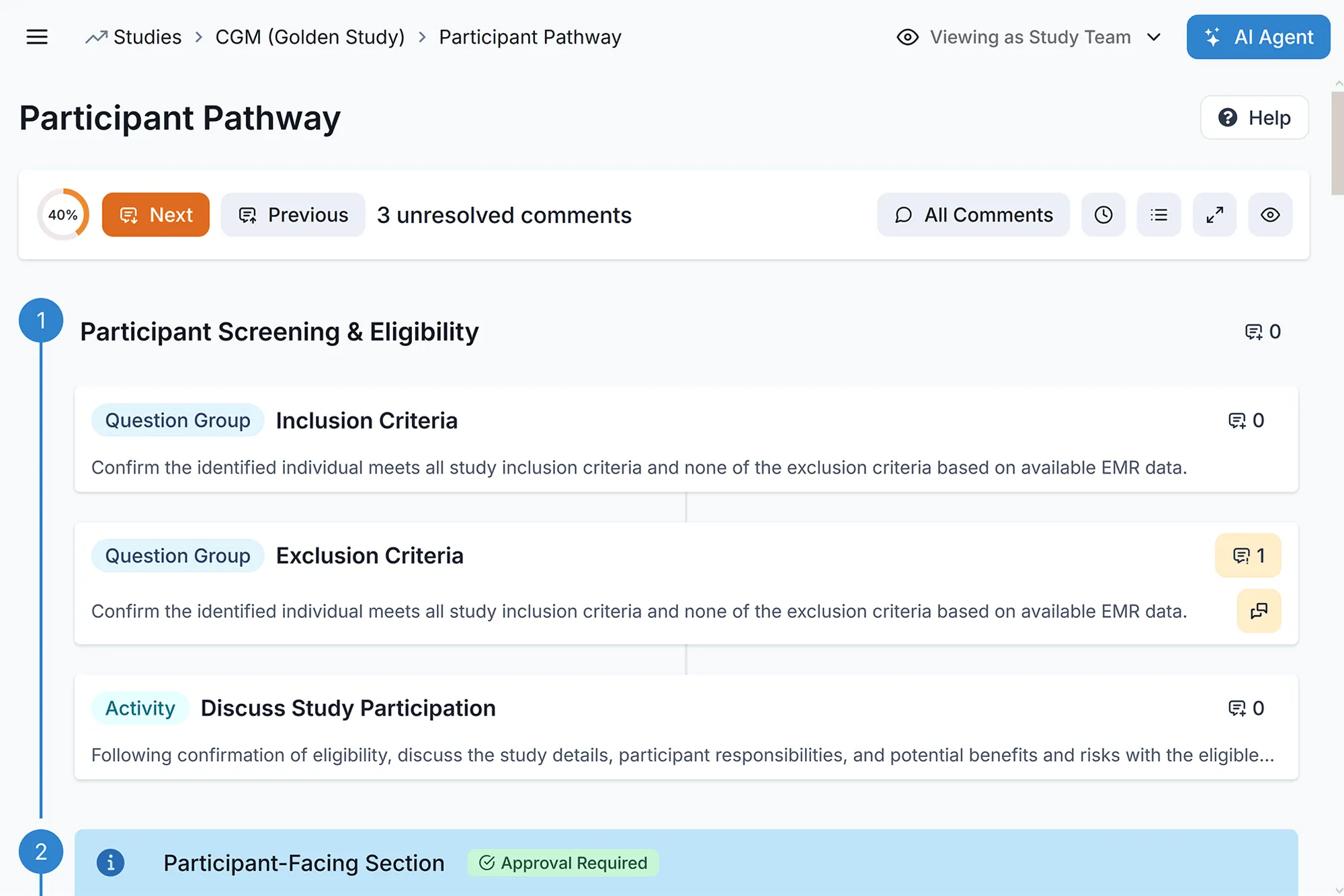
Unified ePRO & EDC Workflows
Operate on a single source of truth. By unifying direct data capture and patient-reported outcomes, your study teams get instant, perfectly synced visibility into patient responses. Streamlined consents and process-first workflows reduce site data entry time from 6 hours to 1 hour per patient.
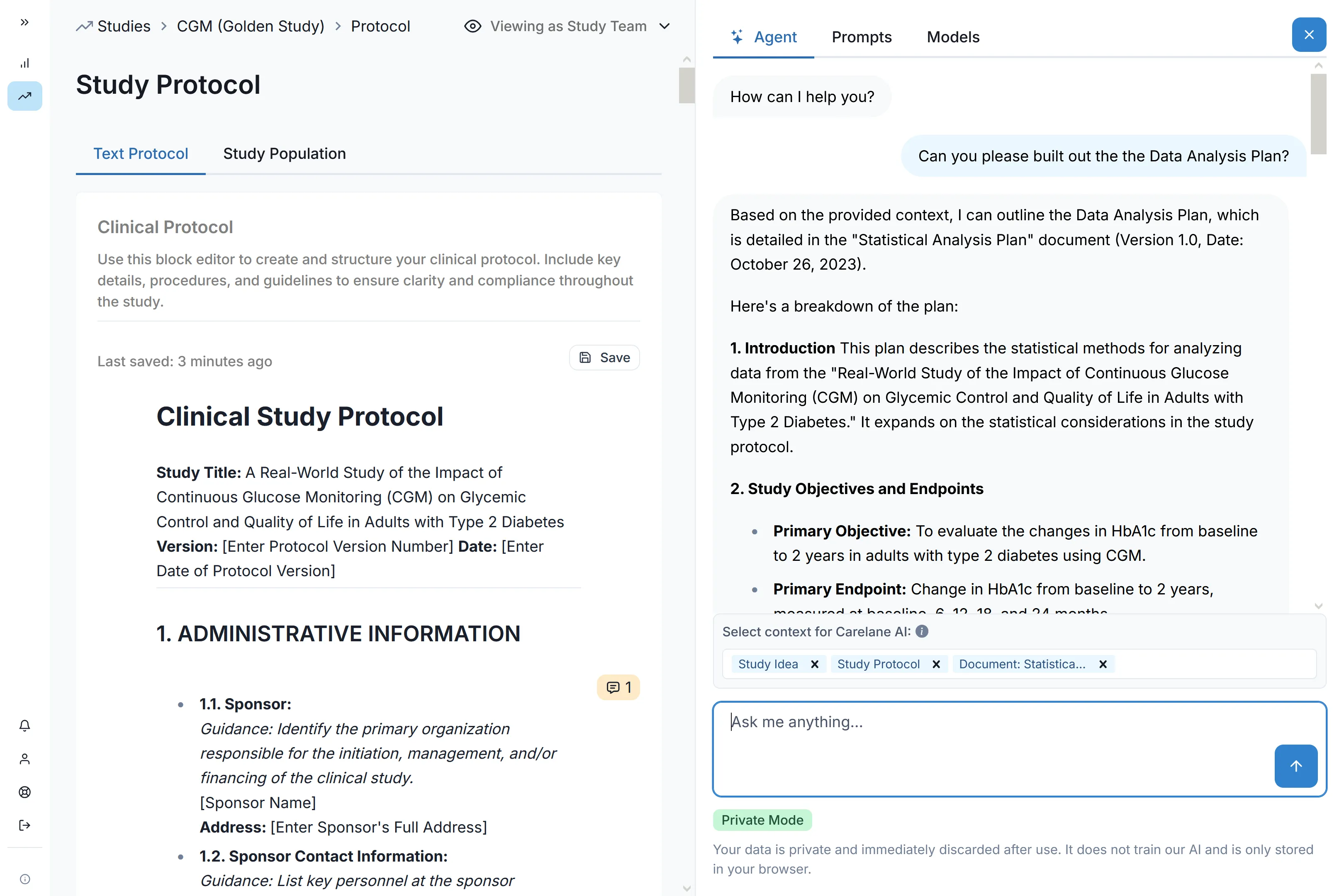
AI-Assisted Query Resolution
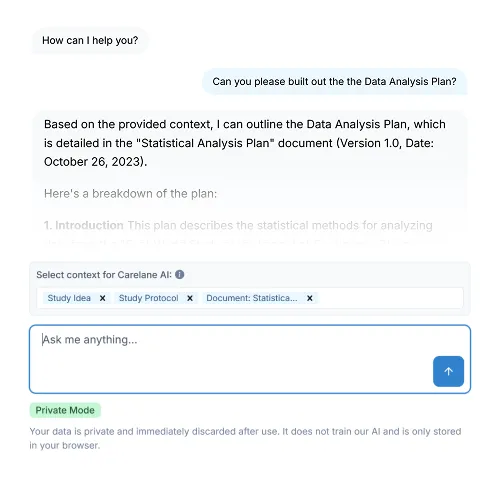
Clean your data as you collect it. Carelane’s intelligent edit checks prevent errors at the point of entry, while AI-assisted resolution workflows reduce query response times from weeks to mere days, drastically accelerating your path to data lock.
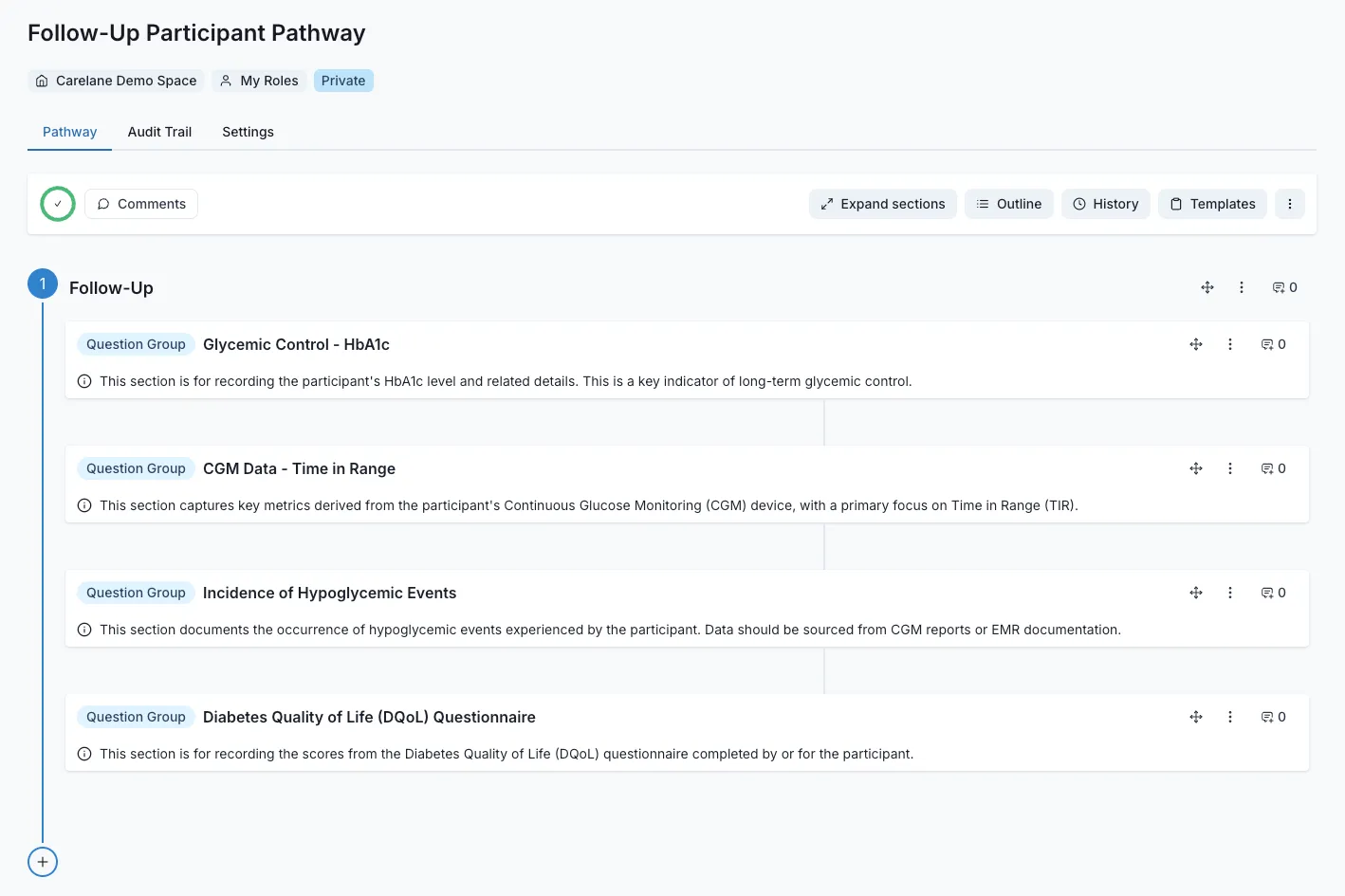
Accelerated Recruitment
Remove the friction that causes patient drop-off before the trial even begins. Our streamlined digital workflows eliminate enrollment bottlenecks. By reducing the administrative burden on practitioners, sponsors double their participant recruitment rates, hitting PoC targets faster.
Proven Momentum for Proof-of-Concept
Sponsors use Carelane to rapidly lock down Phase II data, validate efficacy, and accelerate their path to Phase III pivotal trials.
The Strategic Imperative of Phase II Speed
Phase II is the ultimate proving ground. The faster you can demonstrate safety and efficacy with pristine data, the sooner you secure Phase III funding, regulatory alignment, or lucrative partnerships.
Accelerated Go / No-Go Decisions
Leadership teams cannot wait months for data reconciliation to decide a drug's fate. Unified platforms provide the real-time, clean data necessary to make rapid, confident capital allocation decisions.
Doing More With Less Capital
With biotech funding cycles tightening, efficiency is survival. Carelane's automated workflows reduce total study costs by up to 80%, allowing sponsors to run robust Phase II trials without burning through their runway.
Interoperable Protocols
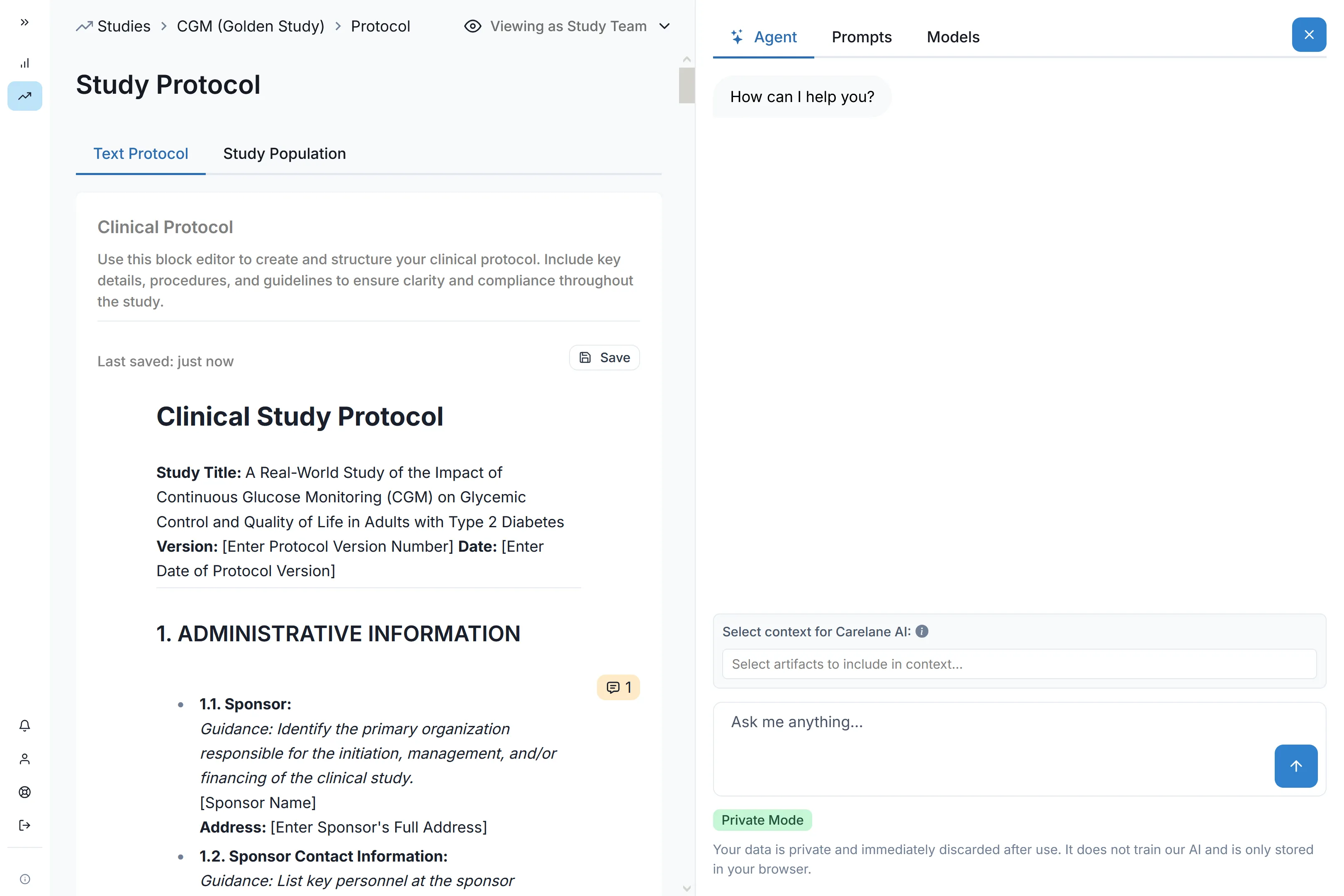
Phase II protocols often require mid-flight adjustments as early data rolls in. Structured FHIR protocols that synchronise automatically across systems mean you can amend your study instantly without breaking the database.
Ready to try Carelane?
The new standard for Sponsors, CROs and Institutions to run faster trials. Instant adoption. Human-in-the-loop AI. Zero compliance risk.