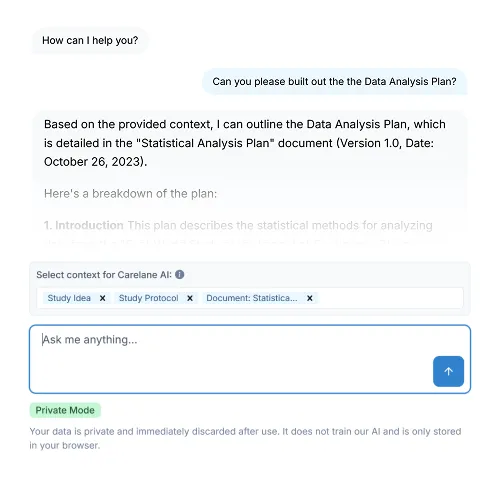
Accelerate First-in-Human and Phase I Trials with Unmatched Speed
Move from protocol to first-patient-in faster than ever. Manage dose escalation, safety monitoring, and complex early-phase adaptive designs on a unified platform built for ultimate speed and precision.
Carelane provides a high-speed, AI-powered platform for First-in-Human and Phase I clinical trials, unifying EDC, ePRO, eCOA and study management into a single source of truth. Designed to eliminate legacy setup delays, Carelane accelerates the path from protocol to first-patient-in by leveraging AI-assisted CRF design and automated dose-escalation tracking.
Why Phase I Trials Suffer from Setup Delays
Early-phase clinical trials require agility and intensive safety monitoring. Yet, rigid legacy infrastructure makes study setup sluggish and protocol amendments painful, delaying critical clinical milestones.
Your early-phase infrastructure should accelerate your milestones, not become the bottleneck.
Speed by Default, Quality by Design for Early Phase
Carelane unifies EDC, ePRO, and study management into a single, high-speed AI-powered infrastructure, enabling you to launch Phase I trials in days and adapt in real-time.

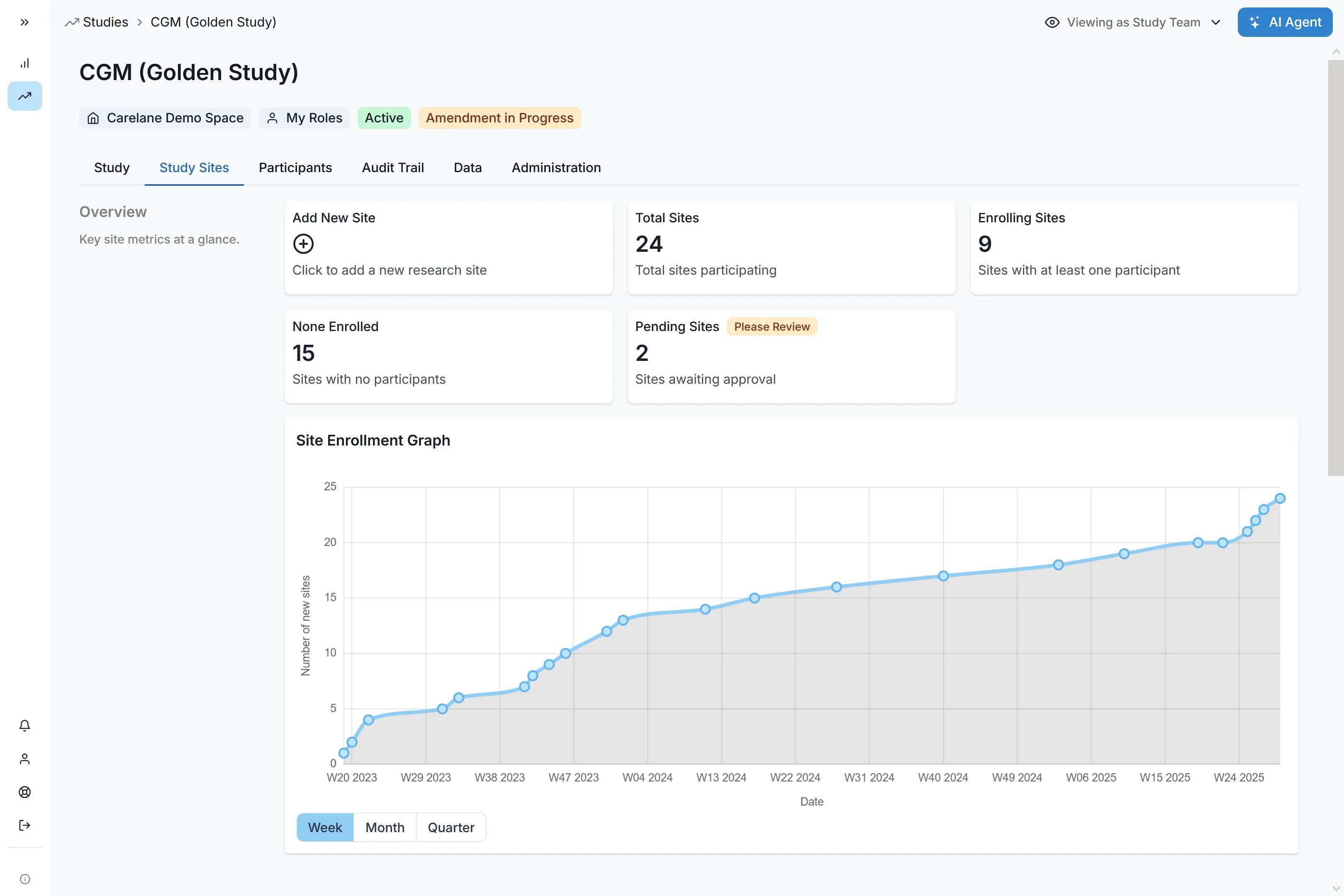
Ultra-Fast Study Setup & Launch
Move from protocol to execution instantly. Carelane's human-in-the-loop AI aligns objectives with outcome measures and leverages pre-validated libraries to reduce CRF design time from 6 weeks to 1 week, ensuring you never miss a milestone window.

Real-Time Safety & Dose Escalation
Equip your safety review committees with immediate data visibility. Seamlessly track patient responses, automate DLT criteria flags, and process data with intelligent edit checks that minimise queries and keep data clean in real-time.
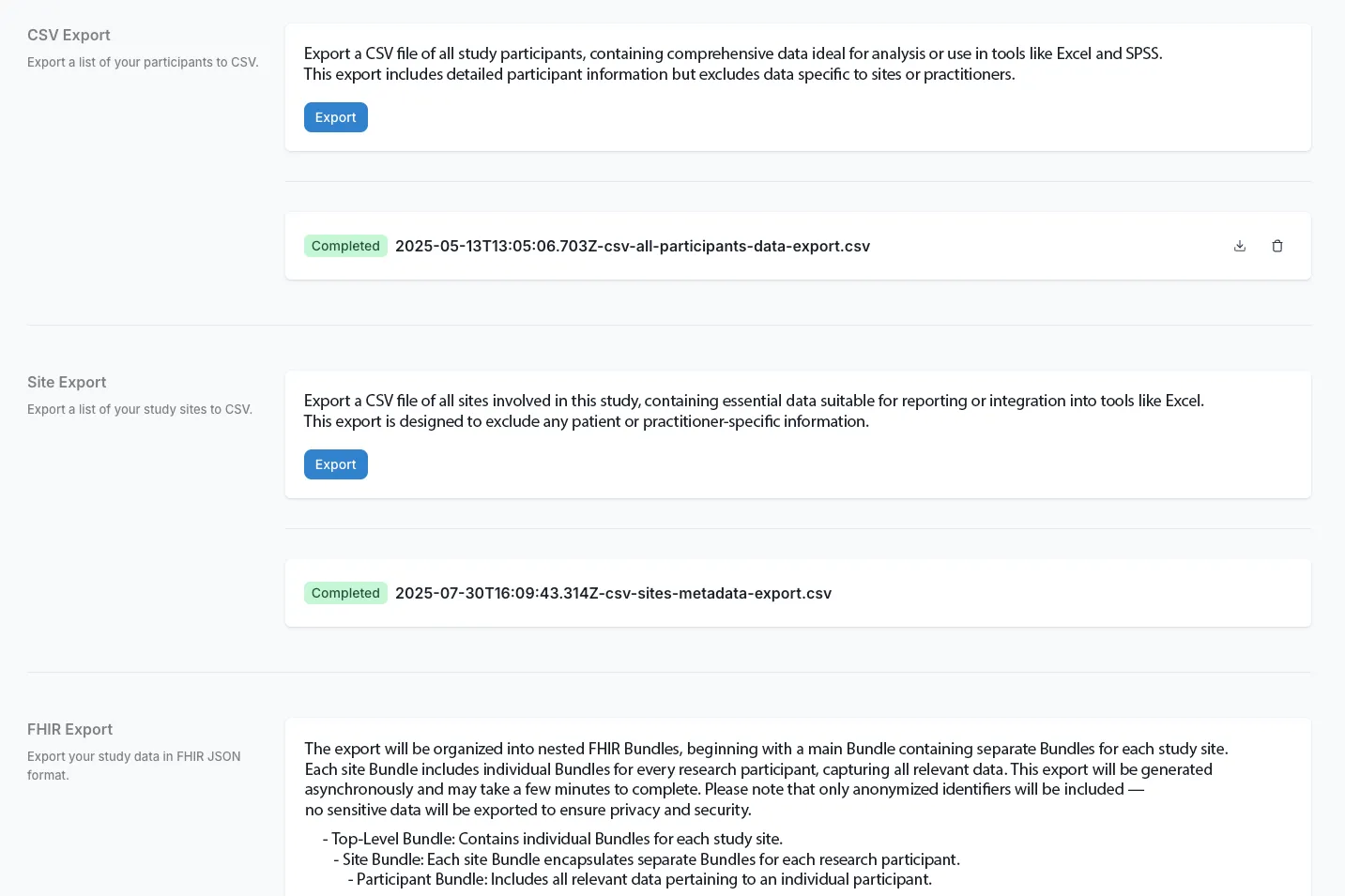
Frictionless Protocol Amendments
Adapt your Phase I design on the fly. Carelane's structured, digital protocol development automatically synchronizes across systems. This accelerates amendments by 50%+ through seamless version control without breaking existing data or delaying site operations.
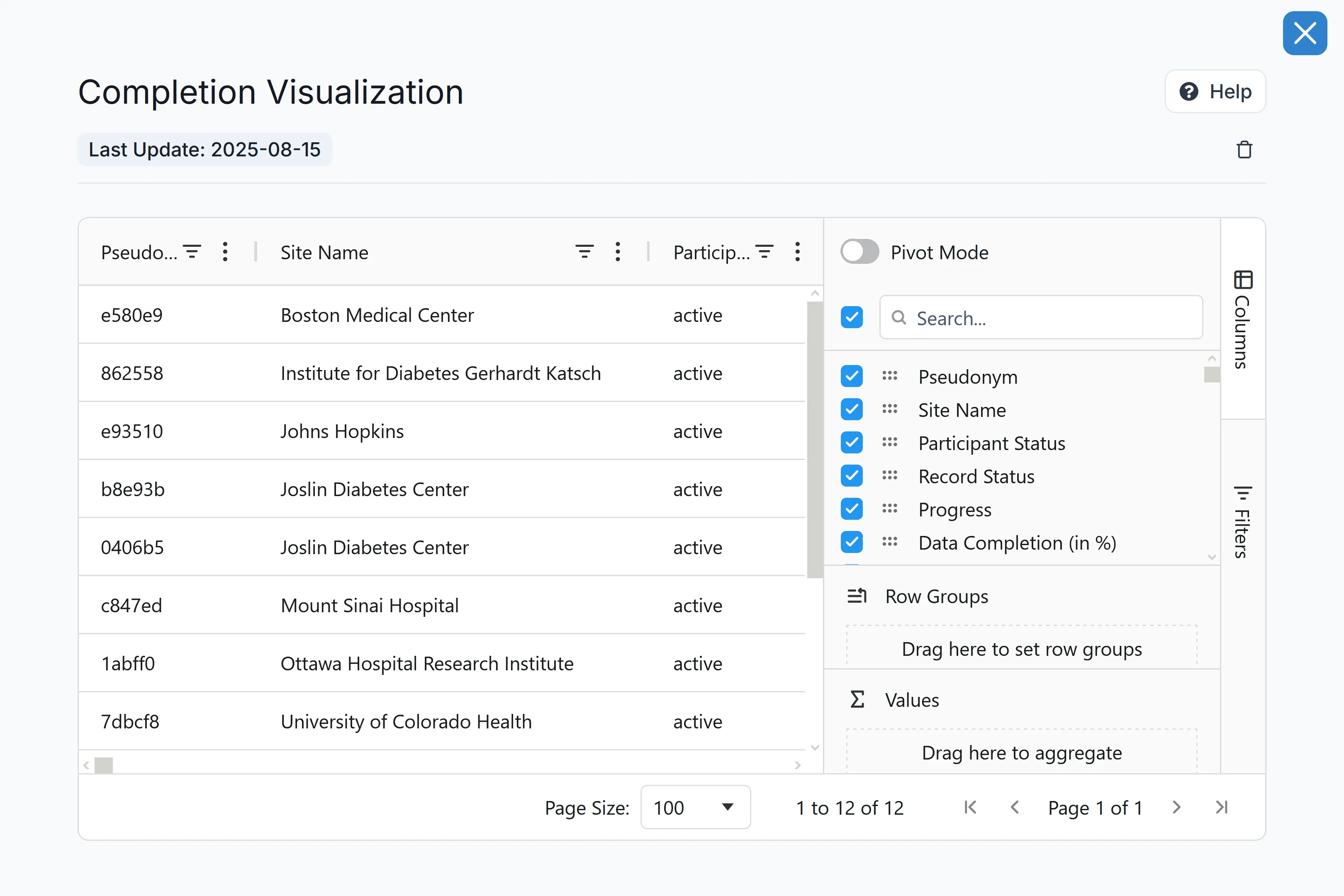
Fortified Security for First-in-Human Data
Protect your most sensitive IP without creating operational bottlenecks. Carelane secures early-phase data with zero-knowledge encryption and architectural data isolation. Combined with precision Role-Based Access Controls (RBAC), your safety review committees get immediate access to the data they need, while unauthorized eyes get nothing.
Speed in Early Phase Execution
Biotech sponsors and Phase I units are leveraging Carelane to safely accelerate their first-in-human trials and achieve clinical milestones faster.
The Strategic Value of Speed in Early Development
In Phase I, time is capital. Sponsors need high-quality data instantly to prove safety, secure funding, and progress to Phase II. Slow infrastructure is a liability you can no longer afford.
Speed to
Milestone
Investors and boards demand rapid execution. The faster you clear Phase I safety and dosage hurdles with clean, reliable data, the faster you unlock your next round of funding or strategic partnerships.
Impenetrable Data Security
First-in-human data is your most valuable asset. With zero-knowledge encryption, architectural data isolation, and precision Role-Based Access Controls (RBAC), your IP and patient PHI remain flawlessly protected.
AI-Driven
Efficiency
Human-in-the-loop AI is becoming the clinical standard. Utilising AI for protocol synchronisation and query management drastically shrinks timelines that used to take weeks down to mere hours.
Ready to try Carelane?
The new standard for Sponsors, CROs and Institutions to run faster trials. Instant adoption. Human-in-the-loop AI. Zero compliance risk.