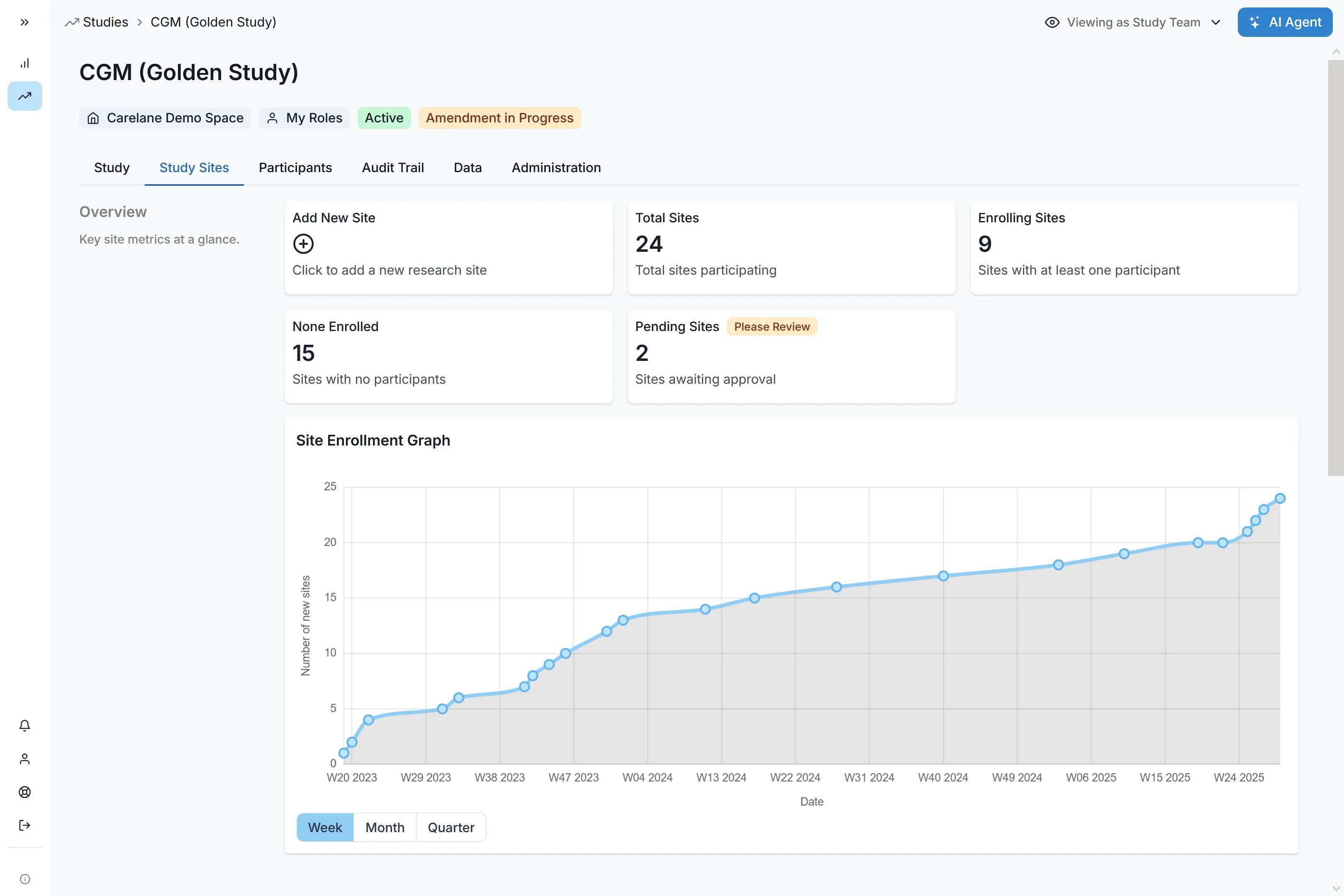
Pricing, finally simple.
Studies/Month
×
Number of Sites
=
Cost
That's the whole formula. No seats, no modules, no surprises.
All features included
Every module, every phase. No tiers.
Generous Al usage
Built for real workloads, not demos.
No hidden limits
Fair-usage policy, no per-seat traps.
One Unified Platform
Built for Phase I-IV Studies, Patient Registries and Academic Studies.
Study Design
- AI-Powered Protocol Digitization
- AI CRF Builder
- Cohort & Endpoint Definition
- Statistical Analysis Plan
- Schedule of Activities
- Ethics/IRB Submission Ready
- Template Management
Study Setup & Conduct
- Site Network Management
- Site Self-Registration
- Full Participant Lifecycle
- Randomisation & Tokenisation
- eSource & Data Capture
- Embedded Protocols
- Seamless Protocol Amendments
Data & Analytics
- Query Management
- Remote Monitoring
- Custom Study Reports
- Data Export
- Reporting & Analytics
- AI Agent
- Full Interoperability
Security & Compliance
- SOC 2 • HIPAA • GDPR
- CFR 21 Part 11
- Granular PHI Encryption
- Role-Based Access Controls
- Continuous Auditing
- Data Isolation & Key Managemet
- Detailed Access & Change Logging
More
General
- Site Onboarding & Training
- Capabilities and Feasibility Workflows
- 2 Factor Authentication
- Encryption in Transit & At Rest
Built for Effective Clinical Research
From CROs to academic labs, one platform delivers faster studies, lower costs, and full data ownership - tailored to how you work.
CRO
Faster study delivery. Lower cost per trial.
- Deliver more studies with fewer resources
- Cut time-to-first-patient with instant site onboarding
- AI-assisted builds replace manual setup
- One unified platform replaces the patchwork of tools
Emerging Biopharma
Own your data. Accelerate to approval.
- Full data sovereignty and portability
- Protocol to submission-ready datasets in one platform
- Compress timelines across every phase by > 70%
- Reach approval faster, at a fraction of legacy EDC cost
Pharma & Institutions
Reduce implementation time from months to days.
- Deploy in days, not months
- Built on natural language - no training cycles needed
- CFR 21 Part 11 and HIPAA compliant out of the box
- Adoption costs a fraction of what you're used to
Academic & IIT Reserarch
Full data sovereignty. Faster time to publish.
- Complete data ownership - fully portable, always yours
- AI-assisted study builds collapse weeks of setup into hours
- Research faster, publish sooner
- No lock-in, no compromises on data control
Ready to try Carelane?
The new standard for Sponsors, CROs and Institutions to run faster trials. Instant adoption. Human-in-the-loop AI. Zero compliance risk.